20/03/2018
- The Microscopic Marathon: Understanding Bacterial Navigation
- Microfluidics: The Precision Tool for Microbial Movement
- Mimicking Nature's Labyrinth: Microfluidics in Bacterial Habitats
- A Single Cell's Story: The Power of Droplet Microfluidics
- The Bacterial Motor: The Engine of Movement
- Classifying Bacterial Taxis: Active versus Passive
- A Spectrum of Responses: Exploring Different Types of Taxis
- Is Bacterial Taxis a Signal Transduction Mechanism?
- The Future of Bacterial Navigation Studies
In the vast, fluid-filled universe of the microscopic, bacteria embark on a continuous journey, driven by an intricate internal compass. This directed movement, known as 'taxis', allows them to navigate their environment, seeking out sustenance, avoiding harm, and ultimately ensuring their survival and propagation. For decades, scientists have been captivated by these tiny travellers, seeking to unravel the complex mechanisms that govern their motility. Traditionally, studying bacterial taxis involved methods like swim plates and capillary assays. While these have provided foundational knowledge, they often present limitations in precision, reproducibility, and the ability to isolate specific environmental influences. However, a revolutionary technology has emerged, poised to transform our understanding: microfluidics. By harnessing the power of precisely engineered microchannels, researchers can now create controlled environments that mimic natural bacterial habitats, offering unprecedented insights into the nuanced world of bacterial navigation.
Microfluidics: The Precision Tool for Microbial Movement
Microfluidic devices are essentially miniature laboratories, crafted with micrometre-level accuracy. Their ability to manipulate fluids in incredibly small channels makes them exceptionally well-suited for studying the behaviour of bacteria, where viscosity, not inertia, dictates movement. The low Reynolds numbers characteristic of these micro-environments allow for laminar flow, meaning fluids move smoothly and predictably, devoid of the turbulent eddies that would obscure precise observation. This control is paramount. It allows scientists to establish finely tuned concentration and temperature gradients, essential for studying phenomena like chemotaxis (response to chemical gradients) and thermotaxis (response to temperature gradients). Furthermore, microfluidics enables the manipulation of other environmental factors, opening doors to understanding rheotaxis (response to fluid flow), magnetotaxis (response to magnetic fields), and phototaxis (response to light). The impact of these advancements is profound, enabling the fine separation of bacterial subpopulations based on subtle differences in their motility traits, with significant implications for synthetic biology, directed evolution, and medical microbiology.
Mimicking Nature's Labyrinth: Microfluidics in Bacterial Habitats
The natural world presents bacteria with a complex tapestry of physical and chemical cues. Microfluidic devices are increasingly being designed to replicate these intricate environments. For instance, microstructures can be fabricated to mimic the porous nature of soil or the cellular architecture within living organisms. This allows researchers to observe how bacteria respond to the continuous hydrodynamic shear forces they encounter in their native settings. The ability to precisely control fluid flow within these devices allows for the generation of tunable conditions that accurately reflect the low-Reynolds number regime experienced by bacteria in their natural habitats. The benefits extend beyond mere replication; microfluidics significantly reduces reagent consumption and assay time, while simultaneously enhancing the reproducibility of experimental results, making the study of bacterial behaviour more efficient and reliable.
A Single Cell's Story: The Power of Droplet Microfluidics
One of the most exciting applications of microfluidics in this field is its capacity for single-cell analysis. By encapsulating individual bacteria within tiny droplets through a process called emulsification, researchers can dissect the behaviour of single cells. This level of detail is crucial, as even within a seemingly uniform population, subtle genetic and phenotypic variations can dictate differences in motility and environmental response. Droplet-based microfluidics provides a powerful platform to differentiate these subtle variations, offering a granular understanding of bacterial behaviour that is simply not achievable with bulk assays. The ability to generate precise and stable gradients of stimuli, such as chemicals, temperature, and pH, within these microfluidic platforms further empowers researchers to conduct highly accurate studies into bacterial taxis.
The Bacterial Motor: The Engine of Movement
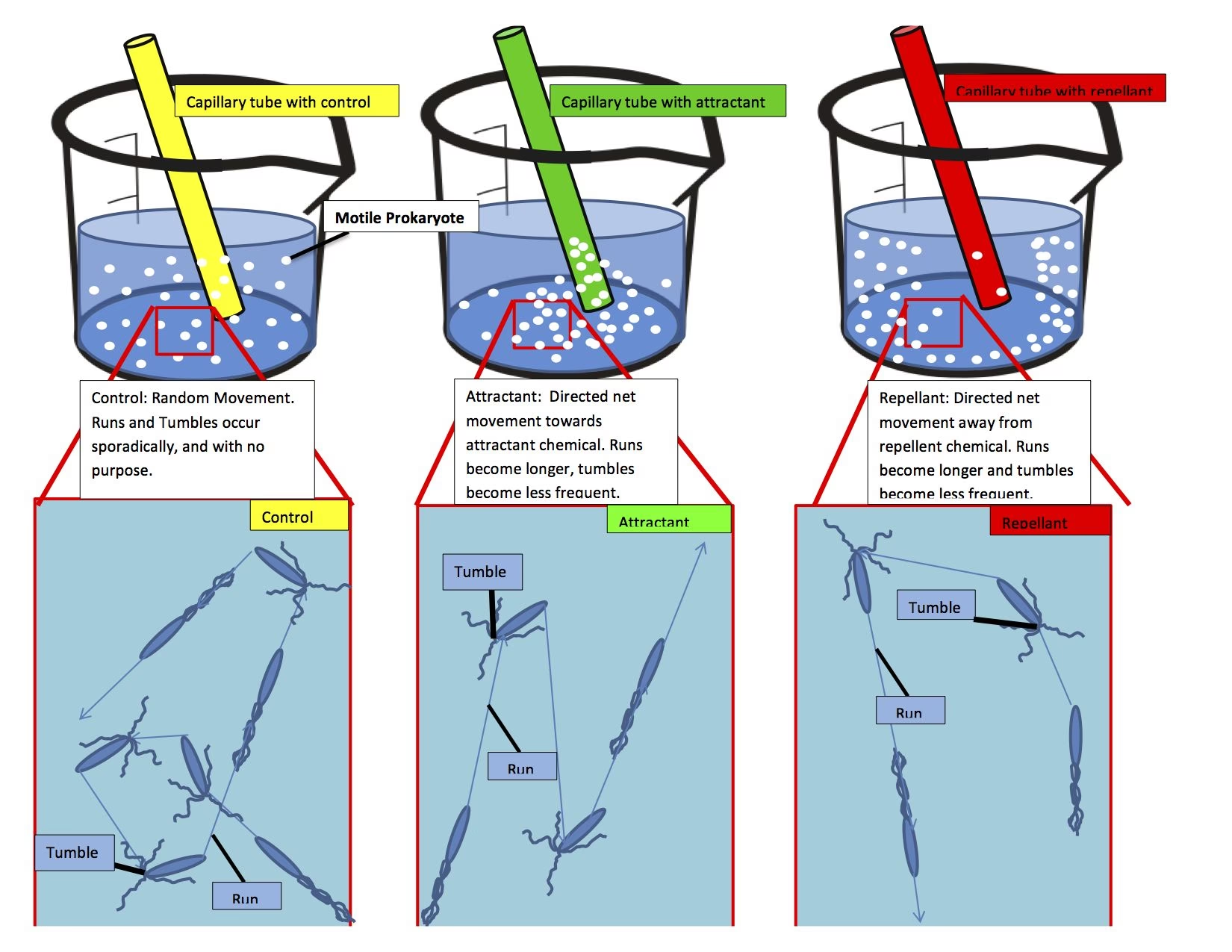
At the heart of most bacterial motility lies a remarkable biological machine: the bacterial flagellar motor (BFM). This transmembrane nanomachine, powered by the influx of ions like protons or sodium, can rotate at astonishing speeds, up to 1000 Hz. The BFM consists of a rotor, to which a long, filamentous appendage called a flagellum is attached, and membrane-bound stator units that generate torque. The magic of bacterial movement lies in the 'run and tumble' dynamic. When all motors rotate counter-clockwise, the flagella bundle together, propelling the bacterium in a straight line – a 'run'. A switch to clockwise rotation causes the flagella to unravel, leading to a random reorientation – a 'tumble'. By controlling the duration of these 'run' and 'tumble' events, bacteria can navigate their environment. This complex process is tightly regulated by internal signalling pathways, such as the phosphorylation of CheY, which influences the probability of rotational switching. This intricate dance of molecular signals and mechanical motion allows bacteria to execute a sophisticated form of 'random walk', optimising their chances of reaching favourable conditions.
Classifying Bacterial Taxis: Active versus Passive
Bacterial taxis can be broadly categorised into two main types: 'active' and 'passive'.
- Active Taxis: In this mode, the bacterium actively modifies its own motility in response to an environmental cue. Examples include chemotaxis, where bacteria move along chemical gradients, and thermotaxis, where they navigate temperature gradients.
- Passive Taxis: Here, an external force is imposed on the bacterium, influencing its movement. Magnetotaxis, where bacteria align with magnetic fields, and gyrotaxis, where they orient in response to gravity and fluid flow, are examples of passive taxis.
Given their microscopic size and low mass, bacteria operate in a low-Reynolds number environment. This means that frictional forces from the surrounding fluid viscosity overwhelmingly dominate over inertial forces. Consequently, maintaining directed movement requires a continuous supply of energy, underscoring the efficiency of their biological motors.
A Spectrum of Responses: Exploring Different Types of Taxis
The ability of biological cells to exhibit directional motility in response to various environmental stimuli is a fundamental aspect of life. While the text focuses on bacteria, it's worth noting that similar principles apply across different organisms, from protists avoiding predators to sperm cells navigating towards eggs. Let's delve into the specific types of taxis studied using microfluidics:
Chemotaxis: The Chemical Compass
Chemotaxis is perhaps the most extensively studied form of bacterial taxis. Bacteria employ sophisticated transmembrane receptor-kinase complexes to sense chemical stimuli. These signals trigger a cascade of molecular events, ultimately regulating the intracellular levels of phosphorylated CheY. This molecule then interacts with the bacterial flagellar motor, influencing the probability of rotational direction changes. During a 'run', bacteria continuously assess the chemical gradient, and if the concentration of an attractant is increasing, they prolong their 'run' time, delaying the next 'tumble'. This allows them to efficiently move towards regions of higher attractant concentration or away from repellents. Interestingly, responses to stimuli like pH and temperature can be 'bidirectional', with bacteria moving towards an optimal condition rather than a monotonic increase or decrease in concentration.
Microfluidic devices play a crucial role in establishing stable chemical gradients for studying chemotaxis. These gradients can be generated through diffusion in static conditions, often within hydrogels or across porous membranes, or through diffusion between parallel laminar flow streams. While laminar flow methods can establish gradients quickly, static hydrogel-based approaches are often simpler to fabricate and prototype. Studies have demonstrated the remarkable sensitivity of this process, with bacteria responding to chemoattractants at concentrations as low as 3.2 nM, a level undetectable by conventional assays.
Rheotaxis: Dancing with the Flow
Rheotaxis is the ability of organisms to sense and respond to fluid flow. In microchannels, a velocity gradient exists, with faster flow in the centre and slower flow near the walls. Bacteria can exploit this gradient, and some species exhibit positive rheotaxis, swimming upstream against the current. Microfluidic devices have been designed to study this phenomenon, observing bacteria swimming in circular paths in still fluid, upstream under moderate flow, and with slight deviation along the flow at high flow rates. The presence of microstructures within these channels can further influence bacterial behaviour, inducing rheotaxis near surfaces and enhancing separation efficiency.
Aerotaxis: The Oxygen Quest
Aerobic bacteria, such as E. coli and Bacillus subtilis, exhibit aerotaxis, moving in response to oxygen concentration. Microfluidic devices capable of generating stable oxygen gradients have been instrumental in studying this. Researchers have observed bacteria accumulating in specific oxygen concentration ranges, indicating a clear preference. High-resolution imaging allows for the precise tracking of individual bacterial movements, revealing their directed migration towards optimal oxygen levels.
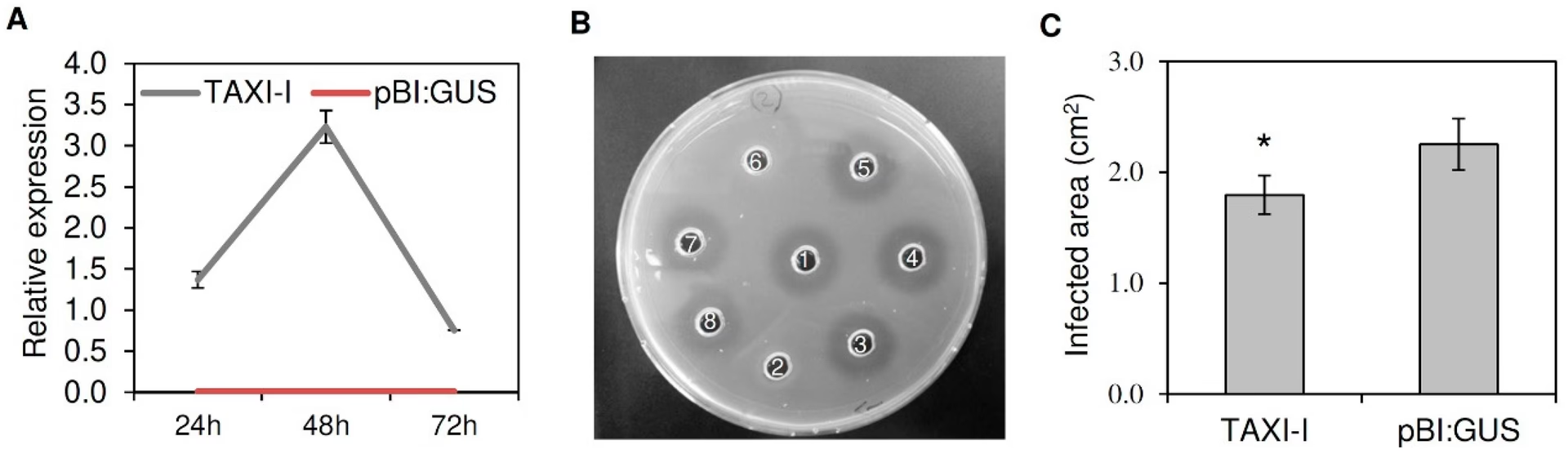
Motile bacteria also exhibit pH taxis, a bidirectional response that drives them towards preferred pH conditions. Microfluidic devices utilizing static hydrogels have been employed to create precise pH gradients. These studies have shown that bacteria are repelled by extreme pH values and accumulate at favourable conditions, confirming the bidirectional nature of this taxis. Furthermore, these devices have allowed researchers to investigate whether microparticle attachment, potentially for drug delivery applications, adversely affects the bacteria's pH sensing mechanisms, finding no significant detrimental impact.
Thermotaxis: Seeking the Optimal Temperature
Thermotaxis, the navigation towards favourable temperatures, is another vital taxis mechanism. Similar to pH taxis, it is bidirectional, with bacteria accumulating at their optimal growth temperature. Microfluidic devices capable of generating both temperature and chemical gradients simultaneously have been developed. These have been used to study the combined effects of these stimuli and to investigate the impact of nanoparticles on bacterial thermotaxis and chemotaxis, revealing that certain nanoparticles can inhibit bacterial migration by affecting essential cellular processes.
Magnetotaxis: Guided by the Magnetic Field
Magnetotactic bacteria possess unique internal structures called magnetosomes, which are ferromagnetic crystals that allow them to align with and navigate along the Earth's magnetic field lines. This phenomenon, known as magnetotaxis, aids them in finding optimal oxygen levels. Microfluidic devices have been used to study this behaviour, observing how magnetotactic bacteria swim along curved surfaces and even alter their direction upon encountering obstacles. The ability to control magnetic fields within these devices also opens up possibilities for targeted drug delivery, where magnetotactic bacteria can be directed to specific cells.
Is Bacterial Taxis a Signal Transduction Mechanism?
Absolutely. Bacterial taxis is, in fact, one of the most extensively studied examples of a signal transduction mechanism. The process begins with the detection of an external stimulus (chemical, thermal, light, etc.) by specific receptors on the bacterial cell surface or within the cell. This detection triggers a series of intracellular events, often involving a cascade of molecular interactions and modifications (like phosphorylation of CheY in chemotaxis). These internal signals are then transduced to the cellular machinery responsible for movement, in this case, the bacterial flagellar motor. The motor's behaviour (run or tumble) is altered based on the strength and type of the signal, leading to a directed movement away from or towards the stimulus. This entire process, from external cue detection to altered motility, is a quintessential example of signal transduction, allowing bacteria to perceive and respond to their ever-changing environment.
The advent of microfluidics has undeniably propelled the field of bacterial taxis research forward. The ability to precisely control the microenvironment, study single cells, and observe complex behaviours with high resolution offers an unparalleled window into the microscopic world. As microfluidic technologies continue to evolve, we can anticipate even more sophisticated designs that mimic natural environments with greater fidelity and enable the study of previously inaccessible phenomena. This ongoing research not only deepens our fundamental understanding of microbial life but also holds immense promise for applications in medicine, biotechnology, and environmental science.
Key Takeaways:
| Taxis Type | Stimulus | Mechanism | Microfluidic Application |
|---|---|---|---|
| Chemotaxis | Chemical Gradients | Sensing chemicals, regulating flagellar motor | Generating precise chemical gradients, single-cell sorting |
| Rheotaxis | Fluid Flow/Shear Stress | Sensing flow direction and velocity | Controlled flow environments, studying upstream swimming |
| Aerotaxis | Oxygen Concentration | Sensing oxygen levels for respiration | Generating stable oxygen gradients |
| pH Taxis | pH Levels | Seeking optimal pH for cellular processes | Creating precise pH gradients, studying bidirectional movement |
| Thermotaxis | Temperature | Seeking optimal temperature for growth | Generating simultaneous thermal and chemical gradients |
| Magnetotaxis | Magnetic Fields | Using magnetosomes for orientation | Applying magnetic fields for bacterial sorting and directed movement |
Frequently Asked Questions:
What is the primary mechanism of bacterial motility?
The primary mechanism is the bacterial flagellar motor (BFM), which rotates flagella to propel the bacterium through a 'run and tumble' motion.
How does microfluidics improve the study of bacterial taxis?
Microfluidics allows for precise control of environmental conditions, creation of stable gradients, single-cell analysis, and higher reproducibility compared to traditional methods.
Can bacteria respond to multiple stimuli simultaneously?
Yes, bacteria can integrate signals from multiple stimuli, and microfluidic devices are being developed to study these complex combined responses.
What are the practical applications of understanding bacterial taxis?
Applications include developing targeted drug delivery systems, improving bioremediation techniques, and advancing synthetic biology.
Is bacterial taxis a form of cellular communication?
While taxis is about individual cell response to the environment, some bacteria use quorum sensing molecules, which are signals, to coordinate group behaviour, which can influence their collective movement or responses.
If you want to read more articles similar to Unveiling Bacterial Navigation: A Microfluidic Marvel, you can visit the Taxis category.

